|
Background info: JJ Thomson used a cathode ray tube in his experiments:
* What is a cathode ray? * What is a cathode ray tube? * What does a cathode ray tube look like? * Just for Fun: What are cathode ray tubes (CRTs) used for today? |
|
Bill Nye Greatest Discoveries: Chemistry Short clip about JJ Thomsons discovery of the electron *watch this as a quick introduction to his experiments |
1st ExperimentThomson put a magnetic field around the cathode ray tube.
- When the ray was bent, no electric charge could be detected at the other end. This meant that the ray itself was ___________________. (charged or uncharged). -Thomson discovered that the cathode ray was made of particles. What were these particles called? |
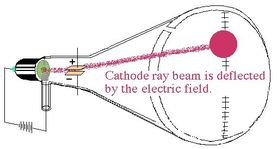
2nd ExperimentThomson put an electric field in the cathode ray tube.
-Towards which charged plate (positive or negative) did the cathode ray bend? -What did this tell Thomson about the electron? |
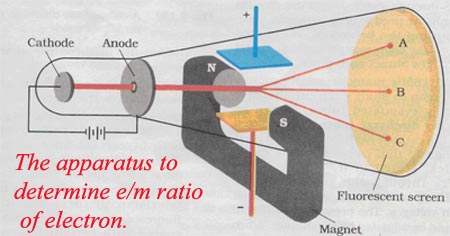
3rd ExperimentThompson designed an experiment using magnetism to move the electrons in the opposite direction caused by the electric field.
- This allowed him to calculate a charge to mass ratio for the electrons. What conclusion about the electron did he make from this data? |
|
Plum Pudding Model |
View the Animation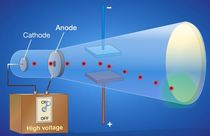
(stop when you get to the equation - you don't need to know it) |
Review:BBC video clip - watch this one if you have extra time or need a quick review NOTE: This is an embedded YouTube video, so if you are watching it at school you may need your teacher to log in for you. |
|